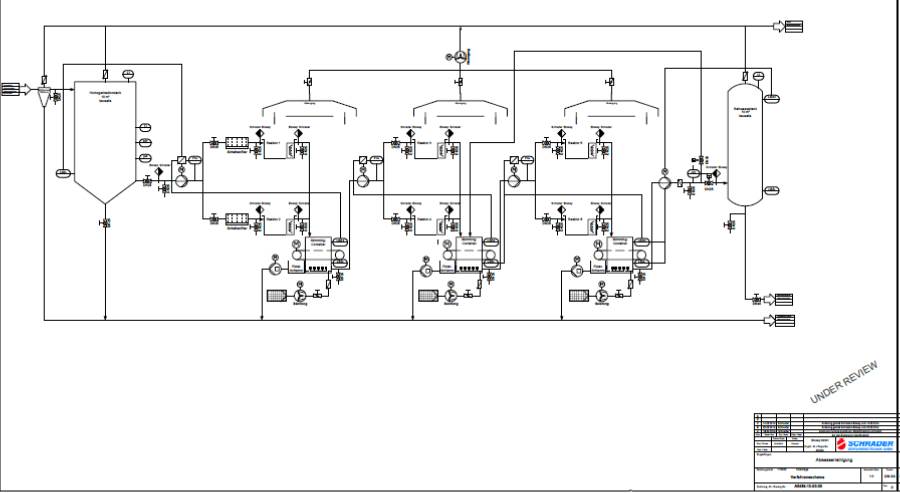
to CO2 and H2O. Same electrochemical reactions occur in stirred and continuous reactors, which are also known as precipitation reaction, metal hydroxides in flake form, to bind the organic impurities. These metal hydroxides are formed by dehydration to metal-oxides with organic contaminates - recyclable so that no residues remain ("0" - waste). These wastewater treatment processes are capable of large mass flows to cope with some 100 m³ / h. They are not subject to any risk of impairment - like biological systems - but are suitable for fully automatic controlled processes. As a by-product, hydrogen is generated at 50 A / 3600s (h) only 0.01141 m³/h which is for recovery of electrical energy too low. 7 V/ 30 A is a standard for EC application (210 VAh) - about 0.78 KWh/m³ is used by Mg-electrodes to form Mg(NH4)PO4 out of waste water. In generall EC needs 1.5 - 2.0 kWh / m³ waste-water.
to CO2 and H2O. Same electrochemical reactions occur in stirred and continuous reactors, which are also known as precipitation reaction, metal hydroxides in flake form, to bind the organic impurities. These metal hydroxides are formed by dehydration to metal-oxides with organic contaminates - recyclable so that no residues remain ("0" - waste). These wastewater treatment processes are capable of large mass flows to cope with some 100 m³ / h. They are not subject to any risk of impairment - like biological systems - but are suitable for fully automatic controlled processes. As a by-product, hydrogen is generated at 50 A / 3600s (h) only 0.01141 m³/h which is for recovery of electrical energy too low. 7 V/ 30 A is a standard for EC application (210 VAh) - about 0.78 KWh/m³ is used by Mg-electrodes to form Mg(NH4)PO4 out of waste water. In generall EC needs 1.5 - 2.0 kWh / m³ waste-water.